Shorter screening time of SARS-CoV-2 enables medical staffs to access patients easier in the emergency room.
Rapiim SARS-CoV-2 Qualitative Rapid Antigen Test System
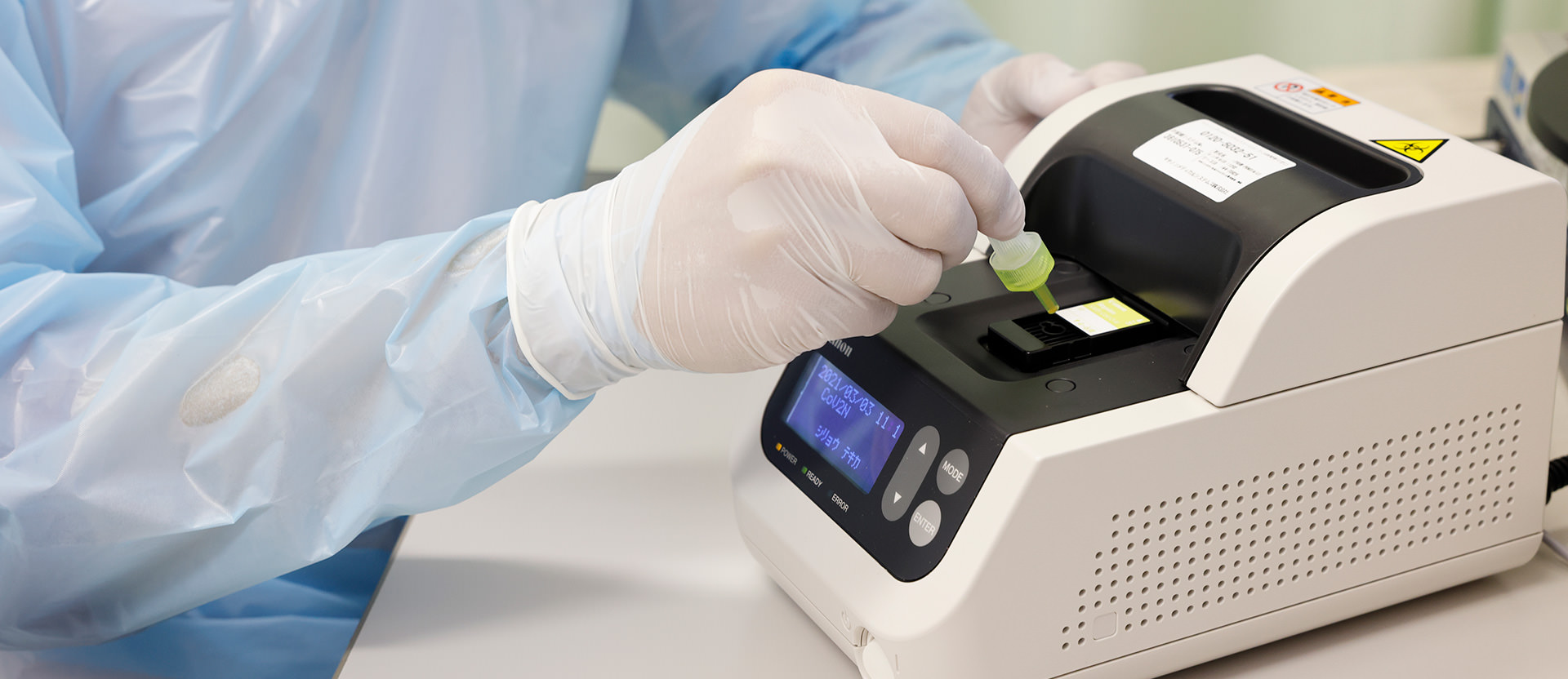
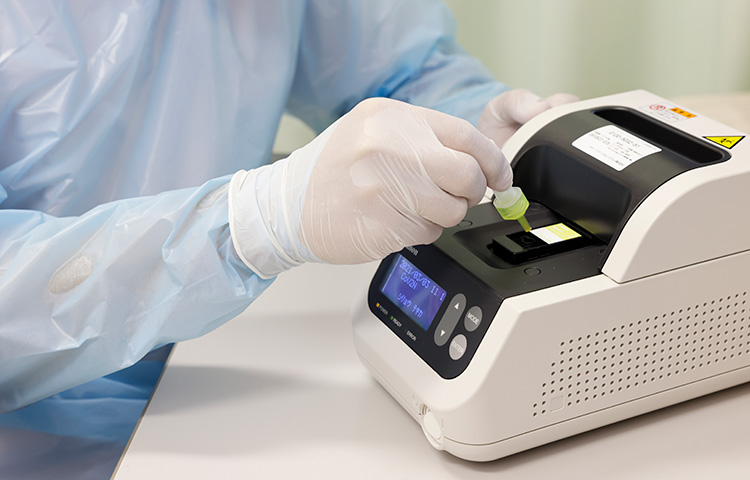
Canon produced rapid antigen test system “Rapiim” which consists of analyzer and dedicated test kits. We have developed a new dedicated test kit for the COVID-19. It takes just as 15 minutes for detecting antigens of SARS-CoV-2*. The test results help medical staffs and improve their working environments in medical institutions.
*SARS-CoV-2 causes COVID-19 after transmitting to people
2022/03/17
Rapid Antigen Test Kit for SARS-CoV-2 Screening
The Coronavirus disease 2019 (COVID-19) pandemic has made hospitals keep guarding against SARS-CoV-2 carriers slipping into their green areas. Rapid tests are helpful for screening SARS-CoV-2 carriers from outpatients. The test kits alarms medical staffs in early stage and they prepare risks and prevent COVID-19 in-hospital infections by isolating the carriers/patients in COVID-19 care units.

Yokohama City University Hospital adopted the rapid test kits co-developed in the collaborative research between Yokohama City University and Canon Medical Systems (called “Canon”)

Dr. Hideaki Kato, Head of Infection Prevention and Control Department, Yokohama City University Hospital
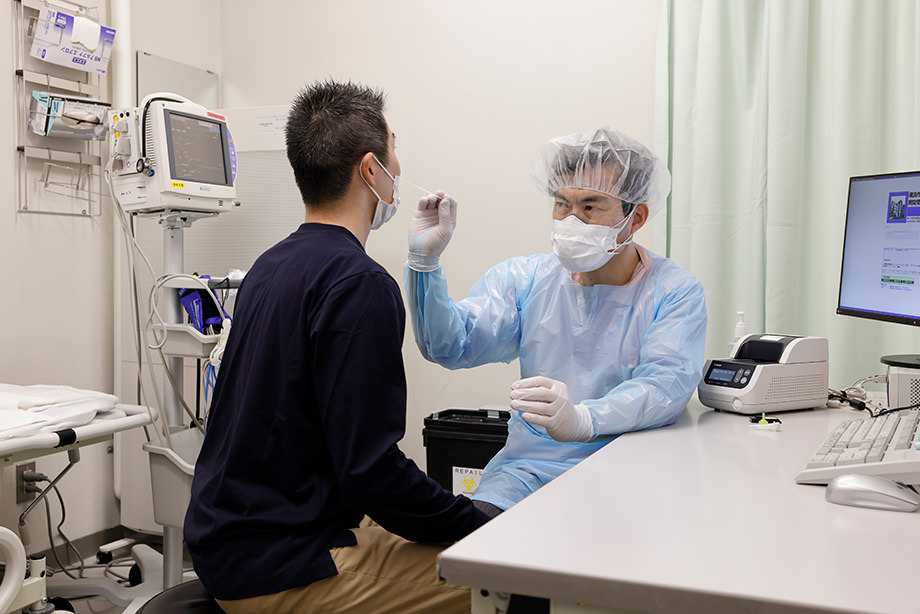
Yokohama City University Hospital admitted COVID-19 patients of first Japanese cluster case of outbreak in cruise ship that entered the Port of Yokohama in February 2020 and has provided medical services for COVID-19 patients. Through our experience in preventing and managing the risk of cluster of COVID-19, we found out that it is necessary to perform screening test for SARS-CoV-2 carriers in short time for managing SARS-CoV-2 positive patients in the facilities. Dr. Hideaki Kato, MD, head of Infection Prevention and Control Department at the university hospital, leads and manages the hospital-wide COVID-19 operations. He started the screening test with immunochromatographic antigen test kits taking half an hour for getting test results at the outpatient fever clinic and emergency room in the hospital.
“As our hospital was originally not a designated medical institution for new infectious diseases, it was very hard to us to separate COVID-19 suspected patient traffic from SARS-CoV-2 negative patient traffic in our emergency room and outpatient fever clinic. We have been performing PCR tests for all patients on admission and patients visiting our emergency room and outpatient fever clinic. PCR tests keep us waiting for half a day, e.g. we take specimen from patients in the morning and get the test results in the afternoon. We must provide medical services to patients as SARS-CoV-2 carrier, with wearing PPE until we get negative results.
Dr. Kato explored alternative new test kits at the point of higher sensitivity and shorter turn-around time. After evaluating available rapid antigen test kits in the market at that time, he adopted Canon’s “Rapiim SARS-CoV-2-N and Rapiim Eye 10” for their screening tests.
A Higher Sensitivity and Specificity Antigen Test Kit
“We initially hesitated for using the conventional immunochromatographic antigen test kits in routine screening, because we heard that other healthcare facilities sometimes encountered false-positive case and false-negative cases with the test kits. We decided to adopt Canon’s Rapiim SARS-CoV-2-N because it was developed in collaborative research between Microbiology Lab at our university and Canon, and it has good performance in sensitivity and specificity.
- *Immunochromatographic test kits: the concept of immunochromatography is combination of chromatography (separation of components of a sample based on differences in their movement through a sorbent) and immunochemical reactions (antigen-antibody reactions). The test kits are simple devices intended to detect the presence or absence of the target analyte. Test results are interpreted visually. The presence of the test line suggests presence of antigen in the kits. The tests are very popular in diagnosis of Flu (Influenza), pregnancy and other respiratory infectious diseases.
The antigen tests for SARS-CoV-2 are designed to diagnose COVID-19 patients with such symptoms as fever (within nine days following onset of symptoms), using a nasal swab (mucus specimen collected from a depth of approximately 2cm) or a nasopharyngeal swab (mucus specimen collected from the back of the nose). These tests make use of immunochemical reaction “antigen-antibody reaction,” an innate immunological mechanism of the human body, to detect target proteins present in the virus (antigens). The Combination of high-sensitive optical detection technology of Canon and high-specific antibody of the Microbiology Lab at the Yokohama City University achieved higher sensitive and specific test kit for detecting COVID-19.
This test system delivers well-balanced performance that satisfies the three key elements in screening of SARS-CoV-2; “higher sensitivity, shorter turnaround time and higher specificity (lower risk of false-positivity due to cross-reaction to other viruses or bacteria).” Additionally, It allows users to test on-site with simple pre-procedures in short time and no-specific facilities such as vacuum chambers.
1. Drop the sample (mixture of reagent and analyte collected from the patient) into the test cartridge
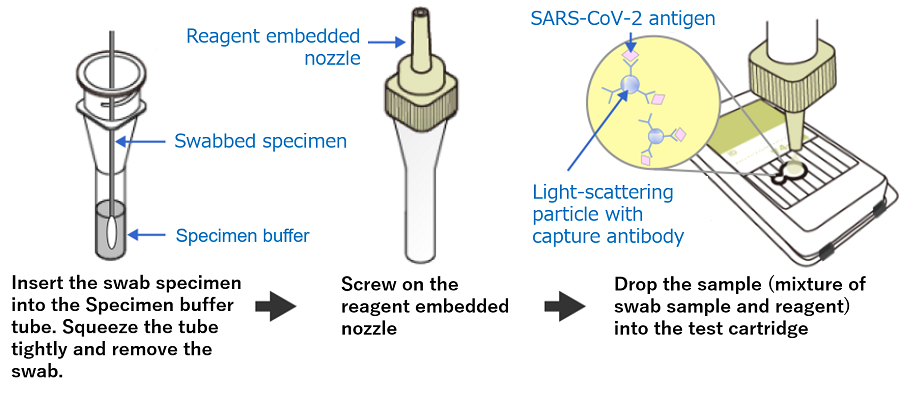
2. Interpretate the data into positive/negative results based on the attenuation of outgoing light from the optical waveguide thin film in the cartridge
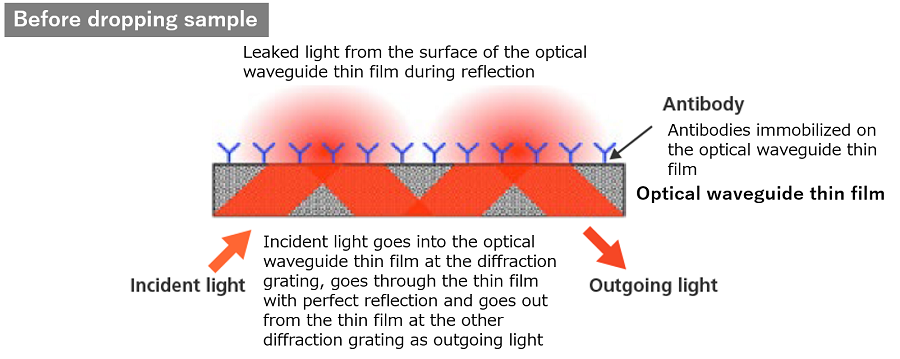
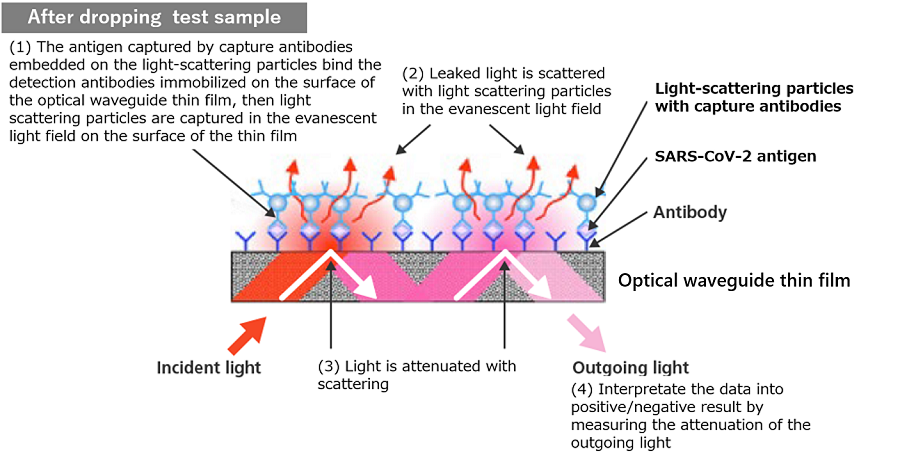
Detection antibodies are immobilized on the surface of the optical waveguide thin film (sensor). Meanwhile, light-scattering particles conjugated with capture antibodies are also prepared to be used for labelling. When antigens are not present, there is no antigen-antibody reaction, and scattering does not occur when light goes through the thin film since the light-scattering particles do not exist in the evanescent light field on the surface of the thin film. On the other hand, when antigens are present, they trigger an antigen-antibody reaction with antigens. Scattering particles are captured in the evanescent light field on the surface of the optical waveguide thin film by antigen-antibody reaction among the antigen and the detection antibodies immobilized on the thin film.
The light is attenuated by the scattering particles at every reflection on the thin film. The analyzer measure the attenuation of the light and translate the value into the results (positive/negative).
Easy Operation at Point of Care
As a doctor working at the front line of medical treatment for COVID-19, Dr. Kato felt that Canon’s test system helps medical staffs because it was small footprint and simple operation.
“In our hospital, the doctors test and diagnose patients with the antigen tests by ourselves. And it is helpful in our operations in outpatient fever clinic and emergency room because of 4 reasons. First, its footprint is so small to install it on a desk in the consultation room. Second, we could test with it everywhere in our hospital because it gets power supply by ordinary outlets (100V, 3Pin) in almost all rooms in our hospital. Third, it boots up so immediately that we start testing in short time. And finally, we could get the results within 4 minutes (minimum) or 15 minutes.
Rapiim soften our stress in providing medical treatment to patients by certifying negative in short time with the test; “We performed Rapiim and chest CT scan on a young patient having symptoms of pneumonia in parallel then we diagnosed the symptom caused by COVID-19 based on CT images and Rapiim results in 30 minutes and started treatment on the patient immediately. It is really stressful for us to continue medical treatment to patients not having certification of COVID-19 with the test. With the antigen test, we could diagnose patients within 30 minutes after patient’s arrival. The rapidity and sensitivity of the test for screening of SARS-CoV-2, would relieve healthcare staff providing treatment to COVID-19 suspected patients.”

Test Softens Stress of Medical Staffs in the Hospital
There are some variabilities in users’ interpretation of visual test line of immunochromatographic antigen test. For example, some users interpretate a faint line as negative, others do the line as positive. Canon’s Rapiim antigen test system shows the interpretated result as “+ (positive)” or “- (negative),” which would make the medical staffs less stressful in interpretation of test results.
Rapiim SARS-CoV-2-N detects SARS-CoV-2 proteins at 6.64pg/ml (LOD: limit of detection) and achieves 5 times higher sensitivity compared with conventional immunochromatographic test devices. Its test procedure is simple (3 steps; make a sample by squeezing the swabbed specimen in buffer solution, drop the sample into the test cartridge set on the analyzer and start analyzing by the analyzer). Additionally, its turn-around time is minimum 4 minutes in case of high density of viral antigens, and 15 minutes in case of no or very tiny density of the antigens. Using the combination of less cross-reactional antibodies in the test kit reduce risks of false positive rate.
“It was very difficult to secure enough clinical laboratory technicians for performing PCR tests especially on weekends, holidays and at night. On the other hand, medical staff could perform the Rapiim without special test skills and get the results very soon. The scientific certification with the test is helpful for convincing all type of staffs in our hospital, and soften their stress in working in next to COVID-19.” said Dr. Kato. In this sense, the higher sensitivity and specificity of the antigen test system helps medical staffs to reduce their stress and worries.
Getting Ready for a Pandemic of a New Infectious Disease
“We feel very happy to see that patients being in isolated room until the day before are discharged without any complication from our hospital”, remarked Dr. Kato. We are preparing to provide medical services throughout prevention and treatment in case of pandemics of new infectious diseases based on the experiences of managing expansion of COVID-19.
Canon will continue to develop products and services in order to promptly provide test solutions that would benefit both patients and medical staffs.
- * This article contains the personal views and comments of the doctor.